12. Exerimental value of magnetic moment of Mn2+ complex is 5.96BM.This indicates a)axial and orbital motion of electron in same direction b)axial and orbital motion of electron in opposite direction c)electron does
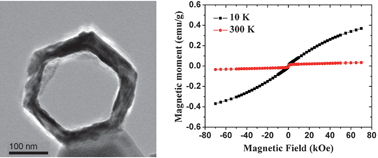
Single-crystal like mesoporous ZnO:Mn2+ nanorings of high optoelectronic quality formed by self-assembly of nanoparticles in an ultrasonic hydrolysis process - Nanoscale (RSC Publishing)

Among V(Z = 23) , Cr(Z = 24) , Mn(Z = 25) and Fe(Z = 26) , which will have the highest magnetic moment?

Mn2+–Mn2+ Magnetic Coupling Effect on Photoluminescence Revealed by Photomagnetism in CsMnCl3 | The Journal of Physical Chemistry Letters
![SOLVED:Calculate the spin-only magnetic moment (Us for the following metal ion. [Note that on the exam you will have to show your work]: High spin Mn2+ SOLVED:Calculate the spin-only magnetic moment (Us for the following metal ion. [Note that on the exam you will have to show your work]: High spin Mn2+](https://cdn.numerade.com/ask_images/bfe7cff006004e7aa567a2395d418232.jpg)
SOLVED:Calculate the spin-only magnetic moment (Us for the following metal ion. [Note that on the exam you will have to show your work]: High spin Mn2+

Which of the following has maximum magnetic moment 1) Fe3+ 2) Mn2+ 3) Mn3+ 4) V3+ - Chemistry - - 14981787 | Meritnation.com

The correct order of spin only magnetic moment (inBM) for Mn^{2+} Cr^{2+} and Ti^{2+} ions is(a} Mn^{2+}>Ti^{2+}>Cr^{2+} (b) Ti^{2+}>Cr^{2+}>Mn^{2+}(c) Mn^{2+}>Cr^{2+}>T^{2+} (d} Cr^{2+}>Ti^{2+}>Mn^{2+} | Snapsolve

Magnetic moment of 2.83 BM is given by which of the following ion? (a) Ti3+ (b) Ni2+ (c) Cr3+ (d) Mn2+ - Brainly.in

calculate the number of unpaired electrons in Ti3+ , Mn2+ and calculate the spin only magnetic - Brainly.in

How many unpaired electrons are present in Mn^(2+) ion ? How do they influence magnetic behaviour of Mn^(2+) ion?


![Solved Mn4+ = [Ar]3d3 Mn2+ = [Ar] 3d5 Mn2+ (HS) Mn2+ (LS) eg | Chegg.com Solved Mn4+ = [Ar]3d3 Mn2+ = [Ar] 3d5 Mn2+ (HS) Mn2+ (LS) eg | Chegg.com](https://media.cheggcdn.com/media/8e7/8e73120c-3a2f-4a30-aaa4-f1ec151322c6/phpFuSNeX)












